
Customize
About Item
Comprehensive training program on Quality Management Systems (QMS) and ISO compliance covering documentation control, internal audits, corrective action processes, risk-based thinking, and continuous improvement frameworks for industrial and processing facilities.
Service Overview
Maintaining consistent quality standards is essential for operational excellence, regulatory compliance, and customer trust. Many facilities struggle with fragmented documentation, unclear quality objectives, weak audit preparation, and inconsistent corrective action processes.
Our Quality Management System (QMS) & ISO Compliance Training program equips facility leaders, quality managers, and operational teams with structured knowledge to implement and maintain robust quality systems aligned with ISO standards and regulatory frameworks.
The training focuses on building systematic processes that enhance product quality, reduce non-conformance, and improve long-term business performance.
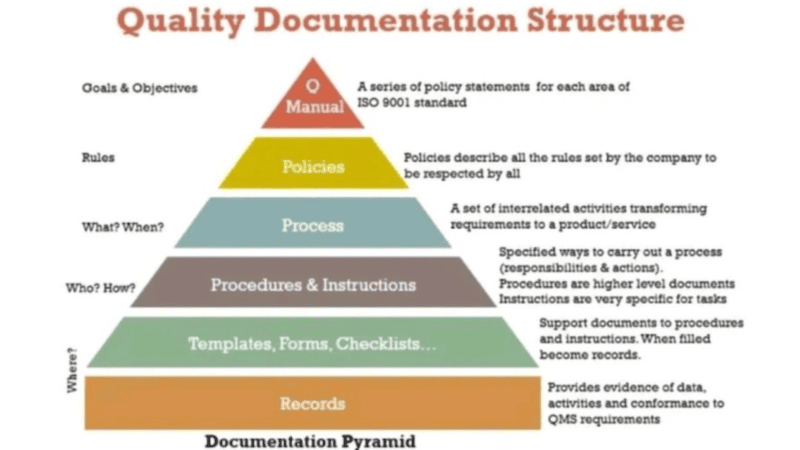
QMS Framework & Documentation Structure
Participants are trained on building a structured QMS framework including:
Quality policy development
Quality objective setting
Process documentation hierarchy
Standard operating procedure alignment
Work instruction documentation
Record control and retention systems
Version control mechanisms
This ensures that quality systems are well-organized and audit-ready.
ISO Standards Awareness & Implementation
The program provides practical guidance on implementing ISO-based systems such as:
ISO 9001 Quality Management principles
Risk-based thinking integration
Process approach methodology
Customer focus alignment
Leadership and accountability frameworks
Continuous improvement models
Participants understand how to align facility operations with global standards.
Internal Audit & Inspection Preparation
Internal audits are critical for identifying gaps before external certification.
Training includes:
Audit planning and scheduling
Checklist preparation
Audit trail verification
Non-conformance identification
Corrective and preventive action planning
Audit reporting documentation
Management review processes
This strengthens audit confidence and compliance readiness.
Corrective Action & Continuous Improvement
Sustainable quality systems depend on improvement cycles.
The program covers:
Root cause analysis methods
Corrective action tracking systems
Preventive action planning
Performance monitoring dashboards
KPI development
Continuous improvement tools
Participants learn how to build improvement-driven cultures.
Risk Management & Process Control
Risk identification reduces operational disruptions.
Training includes:
Operational risk mapping
Quality failure prevention
Supplier risk assessment
Process deviation monitoring
Control point identification
Documentation of risk mitigation strategies
This improves process stability and reliability.
Digital QMS & Automation Tools
Modern facilities use digital quality tools.
Participants are introduced to:
Digital document management systems
Audit management software
Non-conformance tracking tools
Performance dashboards
ERP integration basics
Compliance monitoring automation
This improves efficiency and reduces manual workload.
Target Participants
Quality managers
Compliance officers
Facility supervisors
Production managers
Warehouse quality teams
Industrial entrepreneurs
Audit coordinators
Business & Operational Benefits
Improved product and service quality
Enhanced audit preparedness
Reduced non-conformance incidents
Stronger regulatory compliance
Increased customer trust
Better documentation governance
Operational efficiency improvement
Long-term certification readiness

