


Customize
About Item
Integrated Facility Hygiene & Sterilization Management Program provides end-to-end sanitation planning, sterilization execution, monitoring, documentation, and compliance support across labs, polyhouses, warehouses, cold storages, and agro-processing units.
Overview
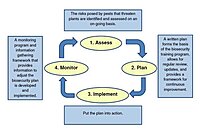
Agricultural facilities often implement hygiene measures in isolated ways—deep cleaning, fumigation, tool sterilization, or water treatment—without a unified framework. This fragmented approach can create gaps in contamination control.
Integrated Facility Hygiene & Sterilization Management Program establishes a comprehensive, facility-wide sanitation strategy. It integrates cleaning schedules, sterilization protocols, biosecurity systems, microbial validation, and documentation into a structured management model.
This ensures continuous hygiene control rather than reactive cleaning.
Facility-Wide Hygiene Assessment
Effective management begins with structured evaluation.
Assessment includes:
Risk mapping of contamination zones
Surface and air microbial baseline testing
Water hygiene assessment
Equipment sterilization review
Entry and exit protocol evaluation
Compliance gap analysis
Detailed assessment builds a customized hygiene plan.
Unified Cleaning & Sterilization Framework
A structured framework ensures consistency.
Program components include:
Zone-based cleaning schedules
Tool and equipment sterilization cycles
Bio-fumigation planning
Air and HVAC sanitation protocols
Irrigation and water tank sterilization plans
Integrated systems eliminate hygiene gaps.
Biosecurity & Access Control Integration
Preventive systems reduce contamination entry.
Integration includes:
Entry sanitation station management
PPE and gowning protocols
Zonal access control
Visitor hygiene guidelines
Staff training modules
Controlled access strengthens protection.
Monitoring, Testing & Validation
Verification ensures effectiveness.
Monitoring includes:
Periodic swab testing
Air quality microbial analysis
Water quality validation
Hygiene audit inspections
Performance benchmarking
Continuous validation maintains standards.
Documentation & Compliance Management
Structured records support regulatory audits.
Support includes:
Cleaning and sterilization logs
Audit-ready hygiene documentation
SOP development and review
Corrective action tracking
Annual hygiene performance reporting
Documentation improves transparency.
Preventive Maintenance & Continuous Improvement
Long-term hygiene requires ongoing oversight.
Program support includes:
Scheduled preventive cleaning cycles
Risk reassessment reviews
Staff refresher training
Process optimization advisory
Sustainability and ESG alignment
Continuous improvement ensures resilience.
Environmental & Operational Benefits
Integrated hygiene management improves reliability.
Benefits include:
Reduced contamination incidents
Lower crop and product losses
Improved compliance performance
Enhanced operational stability
Stronger brand credibility
Unified systems protect revenue streams.
Strategic Importance in Agri Operations
Integrated hygiene management is critical for high-value agriculture, export-oriented operations, biotech labs, and food processing facilities. It ensures contamination control across the entire operational lifecycle.
Strategic advantages include:
Holistic contamination prevention
Improved audit outcomes
Reduced operational disruptions
Long-term cost efficiency
Enhanced sustainability performance
Ideal Customers
Tissue culture laboratories
Polyhouse and greenhouse operators
Agro-processing plants
Cold storage facilities
Post-harvest handling centers
Agricultural research institutions

